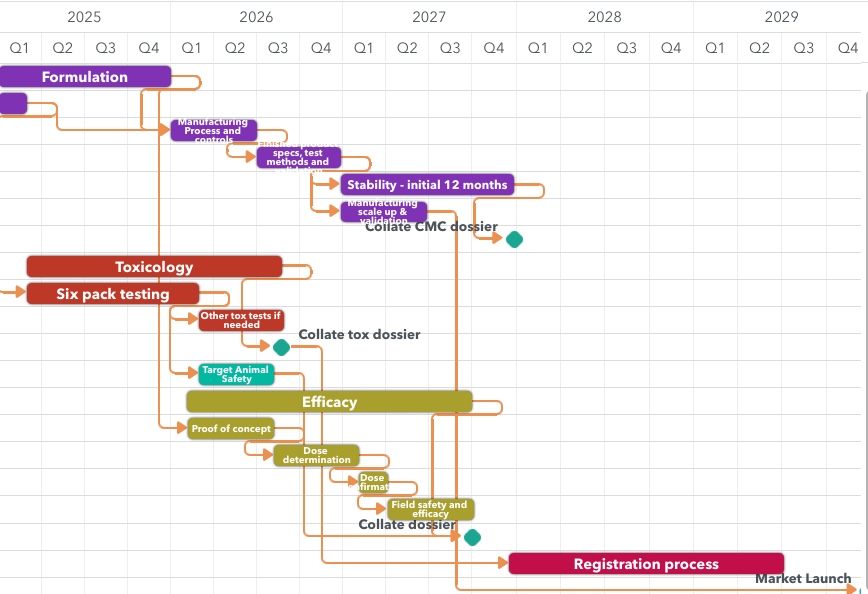
We can guide and support you from start to finish of your product development journey. With our expertise and more than 25 years’ combined experience in registering new products, we can advise on Chemistry and Manufacturing, Efficacy, Safety and Residues requirements, meet with regulators, help you build your registration data package and shepherd it through the assessment and approval process, ensuring your product will be compliant with local regulations.
What We Cover:
- Chemistry and Manufacturing Controls (CMC)
- Efficacy study design and execution
- Safety testing protocols
- Residues analysis and withdrawal periods
- Regulatory authority liaison
- Data package compilation and submission
- Post-approval compliance monitoring
Our Expertise:
- New Zealand and Australian regulatory frameworks
- Veterinary Medicines Directorate (VMD) requirements
- APVMA (Australian Pesticides and Veterinary Medicines Authority) compliance
- International harmonization standards
- Risk assessment and mitigation strategies
Ready to start your product development journey? Learn more about our comprehensive product services.